Introduction: (Initial Observation)
Air pollution is the contamination of the air by noxious gases and minute particles of solid and liquid matter (particulates) in concentrations that endanger health. In addition to many economical and agricultural losses, air pollution is the main cause of many diseases and deaths every year. Excessive growth rate of air pollution is a major concern for many countries and scientists from all over the world are studying on causes, prevention methods and cleanup of the air pollution.

This project is an opportunity to follow the foot steps of other scientists and learn about the air pollution causes and cleanups.
Information Gathering:
Find out about air pollution. Read books, magazines or ask professionals who might know in order to learn about the causes of air pollution and methods of prevention and cleanup. Keep track of where you got your information from.
For basic general information, encyclopedia is a good start.
Air Pollution Control
Purpose
To show how air pollution is controlled.
Grade level
6th, 7th & 8th grades
Essential Elements
(Science) 1 (A) Properly demonstrate the use of laboratory equipment; 2 (A) Observe physical and chemical properties of matter; 5 (A) Measure physical and chemical properties of matter.
Objectives
At the end of the lesson the student will be able to distinguish between an electrostatic precipitator and a wet scrubber and the principles behind the operation of these control techniques.
Focus
When any product is made by industry, waste may be produced that can pollute the air. Wet scrubbers and electrostatic precipitators are two devices used to clean up the air waste stream before it enters the atmosphere.
Background
Air contaminants are emitted into the atmosphere as particulates, aerosols, vapors, or gases. The most common methods of eliminating or reducing pollutants to an acceptable level are destroying the pollutant by thermal or catalytic combustion, changing the pollutant to a less toxic form, or collecting the pollution by use of equipment to prevent its escape into the atmosphere. Pollutant recovery may be accomplished by the use of one or more of the following:
Baghouses – Dry particulates are trapped on filters made of cloth, paper or similar materials. Particles are shaken or blown from the filters down into a collection hopper. Baghouses are used to control air pollutants from steel mills, foundries, and other industrial furnaces and can collect more than 98 percent of the particulates.
Cyclones – Dust-laden gas is whirled very rapidly inside a collector shaped like a cylinder. The swirling motion creates centrifugal forces causing the particles to be thrown against the walls of the cylinder and to drop into a hopper. Cyclones are used for controlling pollutants from cotton gins, rock crushers, and many other industrial processes and can remove up to 95 percent of solid pollutants.
Electrostatic precipitators – By use of static electricity, they attract particles in much the same way that static electricity in clothing picks up small bits of dust and lint. Electrostatic precipitators, 98 to 99 percent effective, are used instead of baghouses when the particles are suspended in very hot gases, such as in emissions from power plants, steel and paper mills, smelters, and cement plants.
Wet scrubbers – Particulates, vapors, and gases are controlled by passing the gas stream through a liquid solution. Scrubbers are used on coal burning power plants, asphalt/concrete plants, and a variety of other facilities that emit sulfur dioxides, hydrogen sulfides, and other gases with a high water solubility. Wet scrubbers are often used for corrosive, acidic, or basic gas streams. ( Note that recovery control devices include adsorption and condenser techniques as well.)
- Which type of air cleaner would be the best for removing particles?
- Which type of air cleaner would be the best for removing waste gases?
- Does a wet scrubber clean up all of the pollutants?
- What problems are produced by having too many pollutants in the air we breathe?
- If industry is just part of the problem, what can we do to control the amount of air pollution that we cause?
Question/ Purpose:
What do you want to find out? Write a statement that describes what you want to do. Use your observations and questions to write the statement.
The purpose of this project is to demonstrate at least one of the air filtration methods. Construct a filter and show that it actually does collect or filter some pollutants.
Possible questions are:
Which filtration method is best for particle pollution?
Which area has the highest amount of invisible pollutants?
What are the causes of air pollution and how can it be prevented? (After identifying the cause of pollution, we can simply stop it by switching to other methods that do not cause pollution. For example if we identify fossil fuels such as coal and oil as a source of pollution, we can try using solar energy, hydroelectric energy or wind energy.)
How effective is any system of air filtration?
Identify Variables:
When you think you know what variables may be involved, think about ways to change one at a time. If you change more than one at a time, you will not know what variable is causing your observation. Sometimes variables are linked and work together to cause something. At first, try to choose variables that you think act independently of each other.
For question 1, variables are:
The independent variable (also known as manipulated variable) is the filtration method.
The dependent variable (also known as responding variable) is the amount of pollutants they filter.
Constants are the type of pollutants and filtration time.
For question 2, variables are:
The independent variable (also known as manipulated variable) is the location.
The dependent variable (also known as responding variable) is the pollution rank.
Constants are the experiment method, time and supplies.
Hypothesis:
Based on your gathered information, make an educated guess about what types of things affect the system you are working with. Identifying variables is necessary before you can make a hypothesis.
Sample Hypothesis:
My hypothesis is that by passing polluted air through water we can filter pollutants and produce clean air. This hypothesis is based on my observation of air freshness after a heavy rain.
Experiment Design:
Design an experiment to test each hypothesis. Make a step-by-step list of what you will do to answer each question. This list is called an experimental procedure. For an experiment to give answers you can trust, it must have a “control.” A control is an additional experimental trial or run. It is a separate experiment, done exactly like the others. The only difference is that no experimental variables are changed. A control is a neutral “reference point” for comparison that allows you to see what changing a variable does by comparing it to not changing anything. Dependable controls are sometimes very hard to develop. They can be the hardest part of a project. Without a control you cannot be sure that changing the variable causes your observations. A series of experiments that includes a control is called a “controlled experiment.”
Experiment 1:
(Visible and Invisible pollutants)
Which area has the highest amount of invisible pollutants?
The atmosphere is almost completely made up of invisible gaseous substances. Most major air pollutants are also invisible, although large amounts of them concentrated in areas such as cities can be see as smog. One often visible air pollutant is particulate matter, especially when the surfaces of buildings and other structures have been exposed to it for long periods of time or when it is present in large amounts. Particulate matter is made up of tiny particles of solid matter and/or droplets of liquid. Natural sources include volcanic ash, pollen, and dust blown by the wind. Coal and oil burned by power plants and industries and diesel fuel burned by many vehicles are the chief sources of man-made particulate pollutants, but not all important sources are large scale. The use of wood in fireplaces and wood-burning stoves also produces significant amounts of particulate matter in localized areas, although the total amounts are much smaller than those from vehicles, power plants, and industries.
In this experiment we will test for visible and invisible pollutants in the air and try to tell the difference between visible and invisible air pollution.
Materials
chart paper
measuring cups
small glass jars
large glass jars
petroleum jelly
3 bean plants approximately the same size
tap water
vinegar
vinegar-water mixture in 1 to 3 ratio
pH paper or indicator
Procedure
Visible Pollutants test
Smear petroleum jelly on each small jar.
Carefully place each small jar inside a large jar.
Decide on several places around the school or home where you think visible pollutants will occur. Make predictions about which area will have more visible pollutants and why. Record predictions in journal.
Place jars in test areas for several days. Check the jars daily. Record observations in journal.
Collect jars for comparison. Observe and rank the jars from the one with the most visible pollutants to the one with the least. Assign each jar a number. Discuss why certain areas have more visible pollutants than others.
Mark a map showing the ranking of areas from the lowest dust to the highest dust.
Invisible Pollutants test
Sets up a bean plant garden with three containers, each container having one bean plant.
Determine and compare the pH of the three solutions and predict how the plants will be affected by each solution. Record pH and predictions in journal.
Plants will be watered every day with 1/8 to 1/4 cup of a solution: one plant with tap water, one plant with straight vinegar, and one plant with the vinegar-water mixture. Procedure is recorded in journal.
Observe plants daily. Record in journal what happens to each plant. Sketches may be part of the observations.
Compare plants and discuss observations at the end of a day, week, two weeks, or until plants die.
Using the observations, write a conclusion for this experiment. Record in journal.
Invisible pollutants are like acid rain. Use the result of your experiment to conclude how does acid rain affect the plants.
Enrichment
Research the history of acid rain. Include information on the causes of acid rain, when we first became aware of the problem, what problems have been caused by acid rain, what measures have been taken to
combat acid rain. Has the situation improved?
Post a chart for the causes of visible pollutants and what can be done to prevent them.
Experiment 2: Make a electrostatic precipitator
Particles (called particulate matter) can be captured before they enter the atmosphere by an electrostatic precipitator. In this experiment we use a plastic tube and black pepper to see how particles are attracted to the sides of the tube much like the pollutants are attracted in large industrial electrostatic precipitators.
Materials, Equipment, and Preparation
plastic tube (fluorescent light tube)
wire coat hanger
plastic grocery bag
electric blow dryer
punch holes, black pepper or rice crispies
Picture on the right shows an industrial model of electrostatic precipitator.
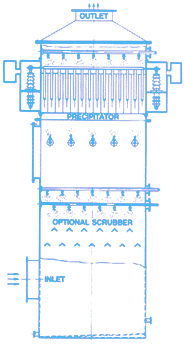
The electrostatic precipitator works on the principle of a static electric charge attracting particles where they are removed.
A 2-foot plastic tube in which fluorescent lights are stored can be used to simulate an electrostatic precipitator. The plastic tube can be charged by running a coat hanger with a plastic grocery bag attached to it.
(The plastic bag as it moves through the tube strips the negatively charged electrons from the inside of the tube making the overall net charge positive. Anything that has a negative charge will be attracted to the tube because opposites attract.)
Hold the tube over some punch holes, black pepper, or rice crispies. Hold an electric hair dryer so the air stream blows across the top of the tube. The air mass creates a low pressure area at the top and the greater air pressure at the bottom pushes the punch holes up the tube. (This is called Bernoulli’s Principal)
***The Results***
If the tube is charged, the punch holes will stick to the sides.
This activity can be used to study static electricity.
If the tube is not charged, the holes will shoot out in a spray.
This activity can be used to study Bernoulli’s principle.
Experiment 3: How to Make a Wet Scrubber
Warning: This experiment requires proper equipment and expert adult supervision. Please skip this experiment without proper equipment and supervision.
The wet scrubber is one of the most common pollution control devices used by industry. It operates on a very simple principle: a polluted gas stream is brought into contact with a liquid so that the pollutants can be absorbed.
In this experiment we will try to build a wet scrubber. (See diagram A)
Materials
Paper towels
12-cm piece of glass
Three 2.5-cm pieces of glass tubing
Three 55-ml flasks
Two glass impingers (glass tubing drawn at one end to give it a smaller diameter so as to let out smaller bubbles)
Heat source (burner or hot plate)
Three 2-hole rubber stoppers (of a size to fit the mouths of the flasks)
Two 30-cm pieces of rubber tubing
Ring stand apparatus
Vacuum source
Procedure
Write your answers on a separate sheet.
Set up the apparatus as shown in attached figure. Put a paper towel in a 55-ml flask and place this above the burner.
Using a 2-hole stopper that makes an air-tight seal with the flask, insert a 12-cm section of glass tubing through one of the holes. The tubing should reach to approximately 1.2-cm from the bottom of the flask.
Insert a 2.5-cm piece of glass tubing into the other hole of the stopper.
Connect a 30-cm piece of rubber tubing to the 2.5-cm piece of glass tubing, making sure an air-tight seal exists.
Fill a second 500-ml flask approximately 3/4 full of water. Using a second double-hole stopper, put a 2.5-cm piece of glass tubing into one of the holes, and insert the glass impinger into the other.
Construct a third flask like the second.
Connect the rubber tubing and heat the first flask (combustion chamber) until smoke appears.
Put a vacuum on the third flask to draw a stream of smoke through the second flask (the wet scrubber). If smoke collects in the second flask above the water, a second scrubber can be added.
Ask the students if particles are the only pollutants produced by industry. Discuss how a wet scrubber collects not only particulate matter but also captures waste gases. Demonstrate how the water scrubber works. Discuss that the white plume you see coming from a smokestack may really be steam coming from a water scrubber.
After observing the wet scrubber, answer the following questions:
Why does the water in the wet-scrubber change color? Why does the wet-scrubber have an impinger (in other words, why is it important for small bubbles to be formed)? What does the scrubber filter out of the air? Not filter out? Suggest ways to dispose of the pollutants that are now trapped in the water.
Materials and Equipment:
List of material can be extracted from the experiment section.
Results of Experiment (Observation):
Experiments are often done in series. A series of experiments can be done by changing one variable a different amount each time. A series of experiments is made up of separate experimental “runs.” During each run you make a measurement of how much the variable affected the system under study. For each run, a different amount of change in the variable is used. This produces a different amount of response in the system. You measure this response, or record data, in a table for this purpose. This is considered “raw data” since it has not been processed or interpreted yet. When raw data gets processed mathematically, for example, it becomes results.
Calculations:
Description
Summery of Results:
Summarize what happened. This can be in the form of a table of processed numerical data, or graphs. It could also be a written statement of what occurred during experiments.
It is from calculations using recorded data that tables and graphs are made. Studying tables and graphs, we can see trends that tell us how different variables cause our observations. Based on these trends, we can draw conclusions about the system under study. These conclusions help us confirm or deny our original hypothesis. Often, mathematical equations can be made from graphs. These equations allow us to predict how a change will affect the system without the need to do additional experiments. Advanced levels of experimental science rely heavily on graphical and mathematical analysis of data. At this level, science becomes even more interesting and powerful.
Conclusion:
Using the trends in your experimental data and your experimental observations, try to answer your original questions. Is your hypothesis correct? Now is the time to pull together what happened, and assess the experiments you did.
Related Questions & Answers:
What you have learned may allow you to answer other questions. Many questions are related. Several new questions may have occurred to you while doing experiments. You may now be able to understand or verify things that you discovered when gathering information for the project. Questions lead to more questions, which lead to additional hypothesis that need to be tested.
Possible Errors:
If you did not observe anything different than what happened with your control, the variable you changed may not affect the system you are investigating. If you did not observe a consistent, reproducible trend in your series of experimental runs there may be experimental errors affecting your results. The first thing to check is how you are making your measurements. Is the measurement method questionable or unreliable? Maybe you are reading a scale incorrectly, or maybe the measuring instrument is working erratically.
If you determine that experimental errors are influencing your results, carefully rethink the design of your experiments. Review each step of the procedure to find sources of potential errors. If possible, have a scientist review the procedure with you. Sometimes the designer of an experiment can miss the obvious.
References:
List of References



